Radon Health Effects
- What is radon?
- How is radon drawn into a building?
- Why is radon a concern?
- Radon is a Lung Cancer Causing Gas
- What happens when radon decay products are inhaled?
- Probability of Radon Induced Lung Cancer
- Scientific Basis for Radon Health Risk Estimates
- EPA and Surgeon General Recommendation
- How does radon rank as a cancer causing agent?
- How does radon compare to other causes of death?
- Radon is a Serious National Health Problem
- Is your area the only area where radon concerns exist?
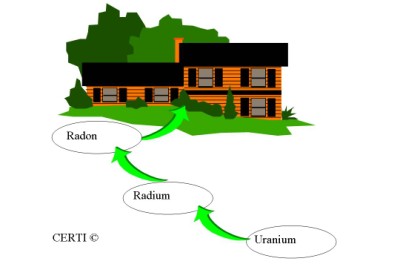
Radon is a gas that is created in the soils where uranium and radium are found. These elements can be found everywhere in the world, therefore any building has the potential for elevated levels of radon. The more uranium found in the soil, the higher the potential for elevated radon levels within a building constructed above this soil. It is not a question of, Is there radon? but rather, How much radon is there?
Radon primarily comes from natural deposits of uranium in the soil. It is not because of a man-made landfill or other suspicious sources. Uranium breaks down to radium, which in turn decays into radon gas.
Radon is an inert gas, which means that it does not react or combine with the elements in the ground. Because of this, radon gas can move up through the soil into the atmosphere, where it is easily diluted and presents little concern. However, when it enters a building constructed on top of this soil, it can build up and become a health concern.
You cannot see or smell radon. There is no way that your body can sense the presence of radon, yet it can have a detrimental effect on the inhabitants by increasing their likelihood of developing lung cancer.
How is radon drawn into a building?
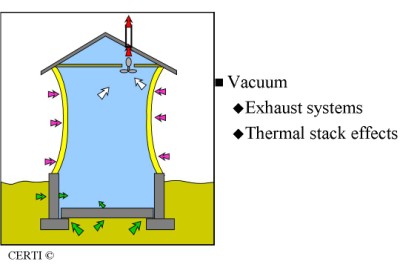 Buildings are typically at a lower pressure than the surrounding air and soil. This causes radon and other soil gases to be drawn into the building.
Buildings are typically at a lower pressure than the surrounding air and soil. This causes radon and other soil gases to be drawn into the building.
There are several reasons why this occurs. One cause is the effect that exhaust fans have when removing air from a building. When air is exhausted, outside air enters the building to replace it. Much of this replacement air comes in from the underlying soil.
When interior temperatures are higher than outside temperatures, thermal effects occur inside of the building. Just as warm air causes a balloon to rise because the surrounding air is cool, warm air rises within a building and is displaced by cold, dense outside air. Some of the outside air, which is displacing and replacing the interior air, comes from the soil.
Whenever air enters a building from underneath, radon will most likely come in as well if radon is present in the underlying soil.
The forces that draw radon into a structure vary, which in turn effects the entry rate of radon. Measurement devices that average radon levels over long periods of time provide a better indication of the amount of exposure to radon.
The minimum duration of any test, the results of which could be used as a basis for determining the need for remediation, is 48 hours.
Radon also varies from season to season as a function of climate and the use of the home by occupants (open windows-closed windows). It would be ideal if a year-long test could be conducted, however, such timing would be unrealistic for the purpose of a real estate transaction.
Short-term tests are utilized to determine the potential of a home for having elevated levels of radon, independently of how the house is operated. The tests involve closing all windows and doors, except for normal exit and entry, and testing on the lowest occupiable location in the home. This technique has become the method for testing homes during real estate transactions.
Once radon enters a building it is easily dispersed through the air. The radioactive decay process that leads to the creation of radon does not stop. This causes the radon to decay into several radioactive elements called radon decay products. These decay products are made up of different forms of polonium, lead and bismuth.
Unlike radon, which is a gas, the radon decay products produced from radon are solid particles. These particles become suspended in the air when they are formed from the decaying radon gas. These particles are extremely small and cannot be seen.
Because they are extremely small particles, radon decay products are easily inhaled and can attach to lung tissue. They have very short “half-lives” which means that they will decay relatively quickly after they are formed. In fact, if they are inhaled, they will decay in the lungs before the lungs have an opportunity to clean themselves.
It is the radon decay products that actually present the health risk associated with radon gas.
Radon is a Lung Cancer Causing Gas
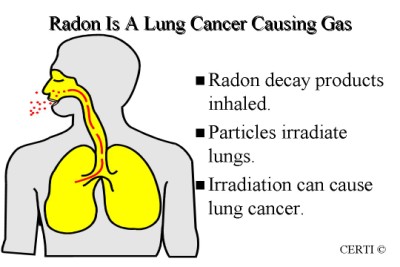 When radon decay products are inhaled they will stick to sensitive lung tissue. Being short-lived, they will break down while they are in the lungs, thereby exposing the lung tissue to radiation.
When radon decay products are inhaled they will stick to sensitive lung tissue. Being short-lived, they will break down while they are in the lungs, thereby exposing the lung tissue to radiation.
When radon decay products decay, they release alpha, beta and gamma radiation. It is actually the alpha radiation (in the form of particles) from the two radon decay products – polonium 218 and polonium 214 – that are the most hazardous.
The alpha particles, that come from the decay of radon decay products, will impact the sensitive lung tissue. In most cases they will kill the lung tissue cell, which can be replaced by the body. However, the alpha particles can impact the DNA, or create a chemical reaction that will affect the DNA. When this occurs the cell can become mutated.
This is the mechanism by which prolonged exposure to radon and radon decay products can increase the potential of lung cancer.
What happens when radon decay products are inhaled?
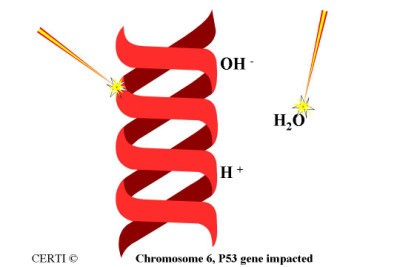 When radon decay products are inhaled they stick to sensitive lung tissue. Being short-lived, they will break down while they are in the lungs. This exposes the lung tissue to radiation.
When radon decay products are inhaled they stick to sensitive lung tissue. Being short-lived, they will break down while they are in the lungs. This exposes the lung tissue to radiation.
If the alpha particle which is released hits a live cell, and within the cell it hits the nucleus, and within the nucleus it hits the DNA, and within the DNA it hits and damages the cancer suppressant gene and increased risk for contracting lung cancer can exist. In addition to the alpha particle causing, the alpha particle can also cause ionization of material around the DNA that can also damage it (shown on right of DNA strand).
This is the mechanism that radon (and more specifically the radon decay products) can lead to an increased risk of lung cancer.
Probability of Radon Induced Lung Cancer
 What is the probability of this happening? It is based on how much you are exposed to and for how long. Being exposed to radon does not mean that you will contract lung cancer. To develop lung cancer from radon the radiation released from its decay has to strike a lung cell and within that lung cell it has to strike a specific location.
What is the probability of this happening? It is based on how much you are exposed to and for how long. Being exposed to radon does not mean that you will contract lung cancer. To develop lung cancer from radon the radiation released from its decay has to strike a lung cell and within that lung cell it has to strike a specific location.
Since the radiation is released in a random manner, the potential for lung cancer increases with exposure, whether that be exposure to a small amount of radon for a long period of time or a high amount of radon for a short period of time.
The probability of contracting lung cancer is like a blind folded person tossing darts at dartboard. If the person tosses a single dart at a time, it is probable that it will take a considerable period of time before the person strikes the bulls eye.
On the other hand, if the person throws a handful of darts at a time, it is likely that less time will pass before the person strikes the bulls eye.
This analogy can be used to understand that the probability of an alpha particle striking a sensitive part of a lung cell in a manner that will cause cancer depends on dose and time of exposure, or Lung cancer risk from radon is a function of:
- Radon concentration to which one is exposed; and
- Amount of time one is exposed to it
Scientific Basis for Radon Health Risk Estimates
The effects of radon have been around a long time. Its effects were first noted as an unknown disease in the 1400’s. Lymphosarcoma was first identified in autopsies conducted in 1879 on European miners.
In modern times (1950’s to present), a higher than expected occurrence of lung cancer has been noted in uranium miners within the U.S., Czechoslovakia, France and Canada. Excess lung cancers have also been observed in other underground (not uranium) miners in Newfoundland, Sweden, Britain, France, China, and the U.S.
The presence of radon at elevated levels in above ground structures, such as homes, commercial buildings and schools was not well known until the mid 1980s. Remember, you cannot see or smell radon. Many homes and schools have been measured with radon levels in excess of underground mine concentrations.
Today the effects of radon are well known. The issue is not if radon causes cancer, but rather, how to control the radon exposure in our homes, workplaces, and schools.
EPA and Surgeon General Recommendation
 Based upon current and past studies, the EPA established a guidance for non-occupational exposures to radon. This guidance level was arrived at by analyzing both the health risks and the cost of fixing buildings with elevated levels. That is, the 4.0 pCi/L guidance is both a health and an economic based number.
Based upon current and past studies, the EPA established a guidance for non-occupational exposures to radon. This guidance level was arrived at by analyzing both the health risks and the cost of fixing buildings with elevated levels. That is, the 4.0 pCi/L guidance is both a health and an economic based number.
The 4.0 pCi/L guidance is not a safety standard. Levels below 4.0 pCi/L still represent some risk. Even the outdoor air contains some radon. So, avoid making comments such as “the house tested safe,” or “there is no radon in the home.” Just because a house is below the EPA’s action level does not mean that there is no risk at all.
The conclusion that radon is a serious health risk is supported not only by the US EPA, but also the American Lung Association, the National Academy of Sciences and the National Council on Radiation Protection, among others.
How does radon rank as a cancer causing agent?
Due to the amount and strength of data that has been collected, radon is placed in the highest category of cancer causing agents. This category is referred to as Group A, which contains materials that are known to cause cancer in humans.
Being a Group A carcinogen certainly underscores the reasons there are concerns about radon. Being a Group A carcinogen also strengthens concerns about liability when a home or commercial building is sold. This is why radon testing has become more common at the time of sale.
How does radon compare to other causes of death?
The comparison above is from the US EPA’s Citizens Guide to Radon. It compares the estimated number of deaths in the U.S. due to radon, with other more widely known causes of death. This comparison provides one reason why the EPA and other health organizations are so concerned about the radon issue. Furthermore, because radon is so easy to detect and also easy to fix, one can see why radon detection and remediation has become such a priority.
The US EPA and the National Academy of Sciences estimate that approximately 11% of the lung cancer deaths in the United States each year can be attributed to radon exposure. This translates to somewhere between 15,000 and 22,000 radon-induced lung cancer deaths each year in the U.S. This includes radon-induced cancer in both smokers and non-smokers.
Applying the U.S. data to the rate of lung cancer deaths in the state of Colorado for the year 2000 and also adjusting for the higher radon exposure in Colorado, it is estimated that 350 lung cancer deaths occur each year in Colorado due to radon exposure.
Notes: per Colorado Dept of Public Health, there were 1,426 lung cancer deaths (LCD) recorded in Colorado during the year 2000. Assuming these were lifetime residents exposed to the Colorado average long-term radon exposure of 2.59 pCi/L, one can factor the U.S. data as follows:
- 18,600 radon LCD (US1995) X 2.59 pCi/L (Colo. Avg.) X 1,426 Colorado LCD (2000) = 350 radon induced Colo. LCD/year
- 157,400 total LCD (US 1995) X 1.25 pCi/L (US average)
Radon is a Serious National Health Problem
The U.S. EPA’s concerns about radon are shared by many other independent health based agencies. During the mid 1980s, the EPA was perceived by some as taking a “Chicken Little” attitude towards radon. That is, they were accused of making too much out of the issue. After all, radon is something that you cannot see or smell. It has been with us for a long time – so what’s the big deal?
However, after several other health based organizations reviewed the data that the National Academy of Sciences used to develop the EPA’s health risk assessments, they joined the EPA as Cooperative Partners to assist in educating the public about this significant environmental health concern.
In addition to the entities listed on the slide, the EPA is joined by many cooperative partners such as the National Association of Counties, The American Lung Association, The National Environmental Health Association.
Is your area the only area where radon concerns exist?
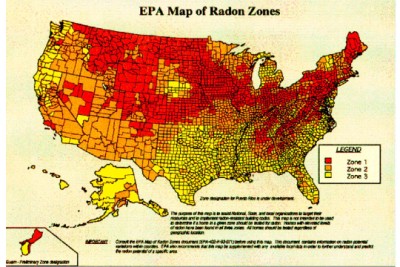 NO! Since the late 1980’s, radon measurements have been taken throughout the United States. This data has been collected and compared to geological formations to yield the map shown above. This map was originally developed for the purpose of establishing the degree of radon reduction methods that should be incorporated into the construction of new homes and buildings. However, it has served as a tool for focusing public awareness efforts on radon. It has also become a tool for relocation companies and lenders to develop their radon testing policies. The zone classifications are based upon being able to predict the likelihood of finding certain ranges of radon concentrations when conducting short-term measurements (as is often done at the time of a real estate transaction).
NO! Since the late 1980’s, radon measurements have been taken throughout the United States. This data has been collected and compared to geological formations to yield the map shown above. This map was originally developed for the purpose of establishing the degree of radon reduction methods that should be incorporated into the construction of new homes and buildings. However, it has served as a tool for focusing public awareness efforts on radon. It has also become a tool for relocation companies and lenders to develop their radon testing policies. The zone classifications are based upon being able to predict the likelihood of finding certain ranges of radon concentrations when conducting short-term measurements (as is often done at the time of a real estate transaction).
Zone 1: Equal to, or greater than, 4.0 pCi/L
Zone 2: Between 2.0 pCi/L and 4.0 pCi/L
Zone 3: Less than 2.0 pCi/L
In comparing zones on the map above, one should remember that the US EPA and Surgeon General recommend that people not be exposed, on a long-term basis, to levels of radon in excess of 4.0 pCi/L. Additionally, one should realize that significant variations can occur within a county and there is no substitute for testing to verify individual conditions.